SANITATION
By Karl Thorson, Food Safety and Sanitation Manager, General Mills and Abby Snyder, Ph.D., Associate Professor, Food Science, Cornell University
In Dry Processing Environments, Treat Water Like Glass
The way we manage glass is the way we should manage moisture in dry and low-moisture food production environments

Image credit: SlavkoSereda/iStock/Getty Images Plus via Getty Images
SCROLL DOWN
In low-moisture food manufacturing, keeping your facility dry is key environmental control. Dry environments restrict microbial growth. Yet many low-moisture food facilities treat water in their environment with a level of complacency that does not match its impact. Poorly controlled humidity levels, water that leaks during clean-in-place (CIP) activities, or condensation on certain surfaces can become routine. The sight of a puddle of water that has always been there stops prompting concern, even though the underlying risk remains the same (Figure 1).
FIGURE 1. Identification of water in the environment of dry facilities should be part of routine environmental controls. The identification of water should be treated with immediate concern. However, too often, water in the environment becomes commonplace even in dry facilities. (Image credit: Cornell Dry Sanitation Research Advisory Council via ChatGPT 5.2)

This complacency is dangerous because water in an otherwise dry food processing environment is like throwing gas on a fire. A small amount of water in these environments will hydrate residues and enable microbial proliferation. In fact, a number of recent U.S. Food and Drug Administration (FDA) letters to low-moisture food processors document persistent water ingress, evidence of inadequate drying, or water accumulating where it does not belong. For example:
- "In several inspections at powdered infant formula manufacturing facilities, FDA observed water present during production in areas that were intended to remain dry (at least during production). The sources of water included leaks from roofs or other exterior facility features, leaks from equipment (during production and/or during sanitation), and condensation. Records of water observed by employees in dry processing areas, and the identified sources, can help a firm analyze trends or identify recurring problems. However, not all firms adequately record this information. The incidence of water in dry production environments should receive prompt consideration by the industry." (FDA Call to Action Letter to the Powdered Infant Formula Industry, March 8, 2023).1
- "On February 17–18, 2022, you found a puddle of water, measuring approximately 4 feet by 8 feet in the bottom of a cooling zone (b)(4) in Roaster (b)(4) and a puddle of water measuring approximately 4 inches by 4 inches in the bottom of cooling zone (b)(4) in Roaster. After you found the puddles of water, you collected environmental swabs from Roaster (b)(4), which were found to be indicative of microbial growth." (FDA Warning Letter: J.M. Smucker LLC, January 24, 2023).2
- "You identified to our Investigator that your cleaning program for the mixing equipment and surrounding clean room includes a wipe down of the equipment with a dry towel, followed by vacuuming, and rinse with clean water. Your Research and Development and Factory manager also identified water without soap is used to rinse mixing equipment after the production of Oat Bran (a product produced on mixing equipment shared with Oat Beta Glucan). Introduction of large amounts of water to clean equipment in a low-moisture food environment can create a moist environment that can support the growth of environmental pathogens such as Salmonella." (FDA Warning Letter: Guangzhou Sinocon Food Co. Ltd., December 23, 2024).3
These findings highlight a common problem. Even in environments designed to remain dry, water has been permitted in, and it is allowed to remain in the environment for too long. What is needed is a more disciplined mindset—one that treats water with the same seriousness applied to other high-risk materials, such as glass.
“Facilities should actively search for water, identify its sources, and remove it with the same rigor expected in a glass control program.”

Glass is avoided wherever possible in food processing areas, and if breakage occurs, the response is immediate. Moisture in a dry plant should be approached the same way. If water is not intentional, controlled, and necessary, then it should be treated as a deviation that demands prompt attention. Facilities should actively search for water, identify its sources, and remove it with the same rigor expected in a glass control program.
Water Dramatically Increases Risk
In a dry facility, risk is reduced when food residues stay below the water activity needed to support microbial growth.4 Once water enters the environment, however—whether intentionally, through wet cleaning or processing, or unintentionally, through a leak or condensation—it immediately changes the equation (Figure 2). Pathogens cannot grow in dry residues or on dry surfaces. Introduce moisture, however, and those same residues become hydrated substrates that support proliferation. Additionally, many pieces of equipment designed for low-moisture food processing are not designed to be wet washed. Consequently, water introduction during sanitation can damage the environment and further contribute to the formation of harborage sites that are harder to remediate once they dry.
FIGURE 2. Pathogen introduction, food soil residue, water, and time are all necessary to support pathogen growth in the environment. Food processors should work to reduce the incidence of each. However, the presence of environmental pathogens is often unknown, and the presence of food residues is often unavoidable. Reducing water and the amount of time when water is present is an actionable step for low-moisture food processors. (Image credit: Cornell Dry Sanitation Research Advisory Council)
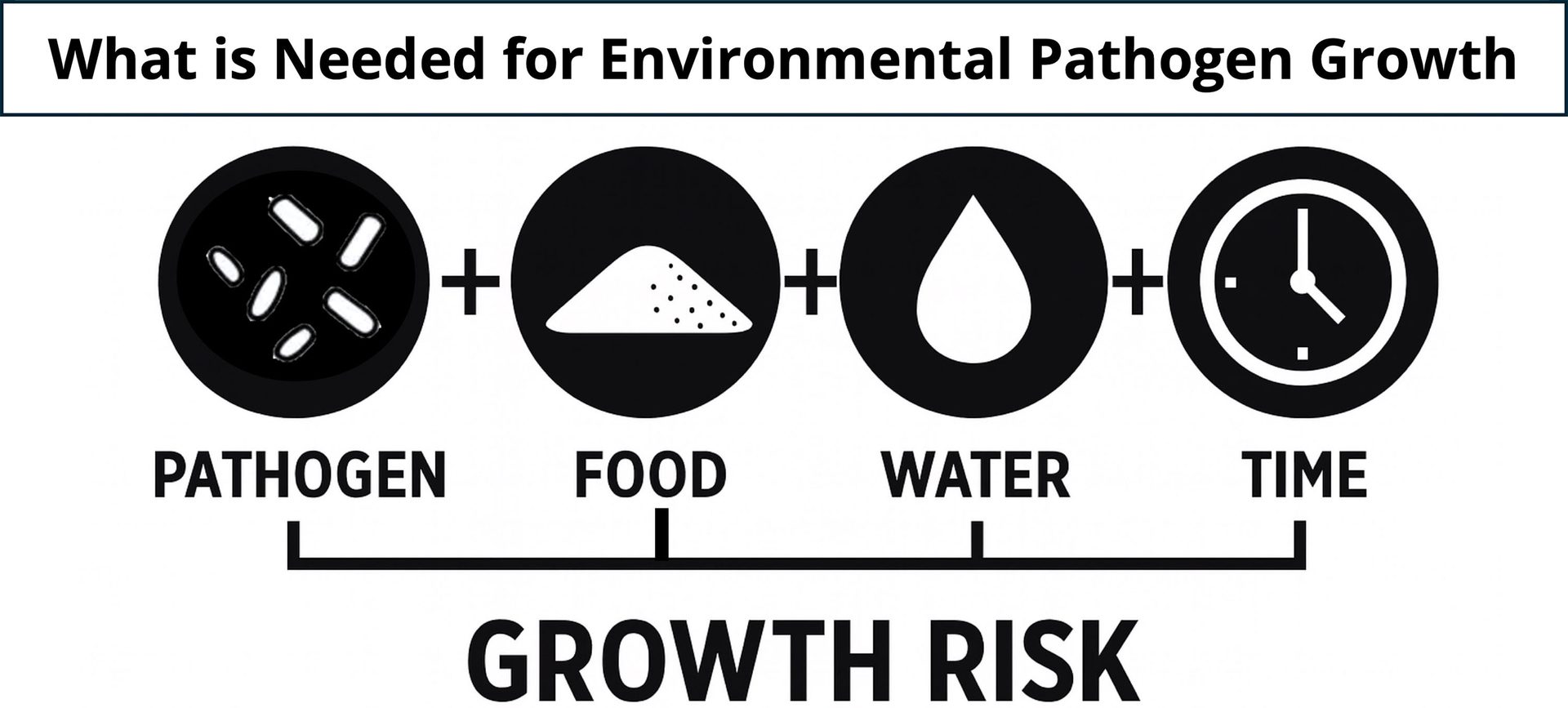
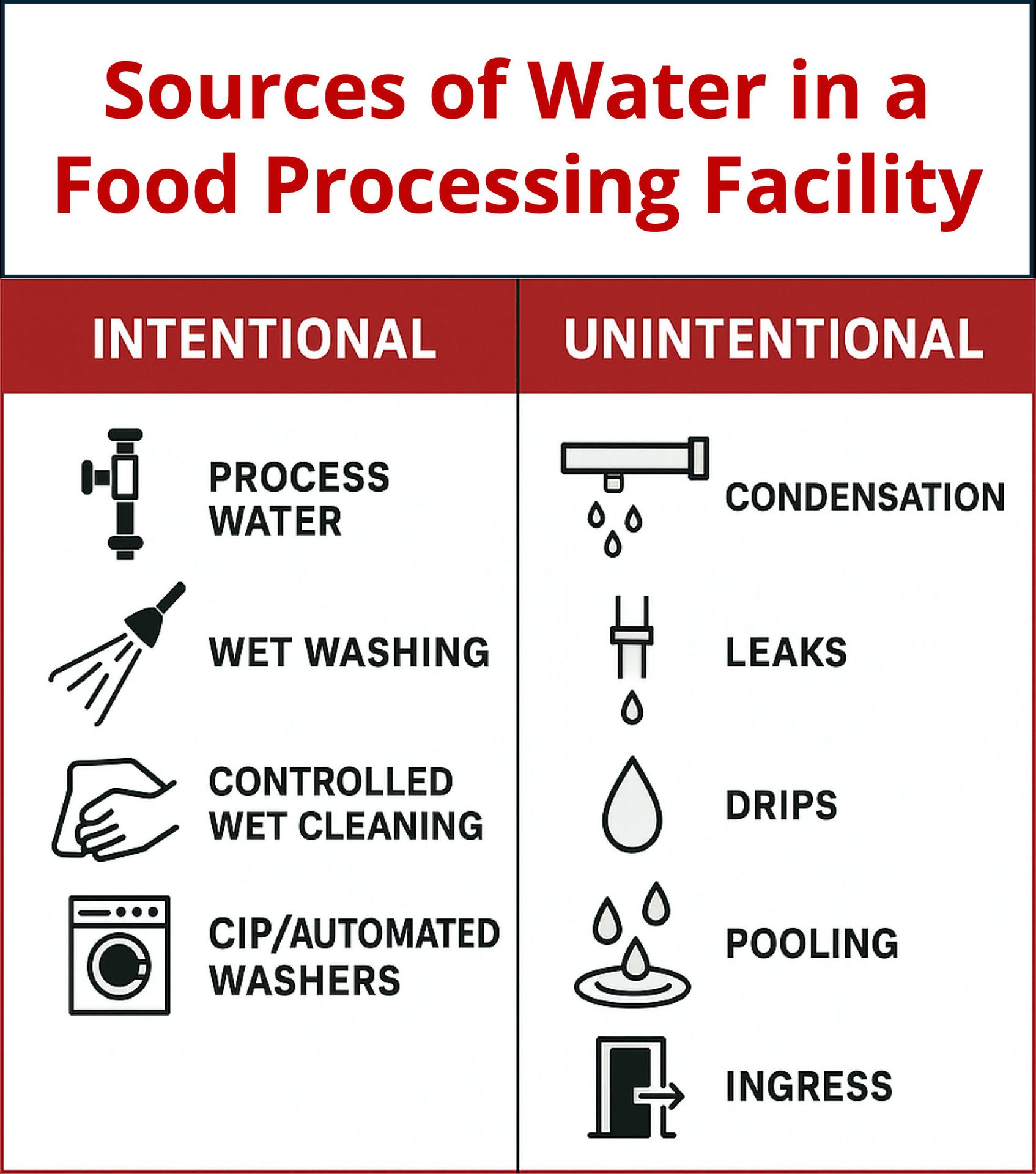
These risks are why the concept of a "war on water" has become common language in dry processing sectors. The program calls for developing a plan for managing water, because uncontrolled water can support substantial growth of pathogens like Listeria and Salmonella.4 Companies report that their war on water programs minimize water usage during cleaning and replace wet cleaning with effective dry cleaning methods wherever practical. Water sources are classified as intentional (process water and essential sanitation) or unintentional (condensation, leaks, drain backups, or imprudent use). Necessary water should be minimized or managed, and unintentional water should be eliminated by fixing root causes (Figure 3). Detailed response actions based on risk, such as shutting down lines, isolating leaks, and documenting corrective actions, are part of the program.
FIGURE 3. Water introduction can occur from a variety of sources. Water control programs should address both intentional and unintentional sources. (Image credit: Cornell Dry Sanitation Research Advisory Council)
Borrowing from Glass Control
One of the best ways to operationalize the "treat water like glass" mindset is to borrow directly from established glass control programs. Food safety programs require manufacturers to prioritize the prevention and control of glass and brittle plastic in their food production areas. The basis of these programs is a comprehensive glass register, a complete inventory of all glass and brittle plastics in production areas (e.g., light covers, clocks, gauge covers, windows, and packaging). This inventory is maintained and audited regularly by staff.
A similar structure can be applied to water. A water register documents all intentional and unintentional sources of moisture along with their locations and potential risks. As glass control programs aim to eliminate or contain glass, a water control program should eliminate unnecessary water sources (e.g., by switching from wet to dry cleaning) or define protective measures such as improved dehumidification systems. Water sources that cannot be eliminated must be managed through routine integrity checks. These could include frequent inspections to verify that seals, insulation, and drains remain intact and that condensation or pooling have not occurred. For example, a Water Audit Checklist for Low-Moisture Food Facilities was originally developed by the Grocery Manufacturers Association (now the Consumer Brands Association) and is available for download from the Snyder Lab at Cornell University website.5
Another critical part of glass control is the contingency plan for breakage. When glass breaks, the area is quarantined, and trained staff clean and inspect. A water control program should apply the same approach. If water is identified, then the source must be isolated, sanitation staff must dry the area, and corrective actions must be documented. A root cause analysis should be conducted to prevent recurrence, and records similar to glass breakage logs should capture the location, cause, remedial actions, and verification results.
Monitoring and verification are also essential. In a glass control program, facilities audit the condition of glass items at scheduled intervals and maintain records. A robust water control program would include moisture inspections, humidity monitoring, and leak detection. Audit frequencies should reflect risk, with high-risk zones receiving more frequent checks. Staff training is needed for those tasked with completion of water control audits, and all personnel must learn to recognize water as a high-impact attribute of their environment and to respond with the same discipline applied to glass.
By mirroring the structure of glass control programs by including a documented inventory, elimination or shielding, contingency plans, routine inspections, and training, a facility can implement a practical water control program that is as disciplined and measurable as its glass control program. This not only reduces microbiological and operational risks but also aligns with regulatory expectations for low-moisture food facilities.
“Facilities often underestimate the cumulative impact of small, repeated introduction of water.”

Water Should Be Introduced Only by Exception
A clear message in the war on water is that water should be introduced only when absolutely necessary. Alternative methods that do not introduce water include purging and manual dry cleaning. These approaches preserve dry environmental conditions and product safety. Water-based cleaning should be used only when necessary and with a clear plan for containment and drying. If a facility identifies a clear and defensible rationale for deploying wet sanitation, then the speed and completeness of dry-out procedures become even more important.
Much of the water that causes problems in dry facilities is preventable. Unnecessary water like condensation, leaks, drips, ingress, and drain failures should simply be eliminated. The purpose of conducting regular water audits is to prevent unnecessary water introduction in the future, not just to identify and correct water ingress during the audit. Seemingly small water sources can support microbial growth or spread contamination across the environment. Facilities often underestimate the cumulative impact of small, repeated introduction of water. A slow drip, condensation on a duct, or a minor roof leak may appear insignificant yet may appreciably enhance microbial risk.
Environmental relative humidity (RH) is a related and highly influential factor that affects a facility's ability to maintain an adequately dry environment.4 It is often overlooked as a critical environmental control, however. Elevated RH increases condensation risk, slows drying, promotes microbial growth over time, and complicates powder handling. Effective humidity management through HVAC, airflow control, dehumidification, and pressure balancing should be understood as a critical food safety barrier in dry facilities.
Engineering controls are powerful tools for controlling moisture. Human diligence matters, of course, but it cannot compensate for facilities that are inherently prone to leaks, condensation, and regular application of wet sanitation. When the physical environment is designed to stay dry, the system is likely to remain dry. That means prioritizing structural and mechanical solutions like properly sealed and insulated panels that block humid air, HVAC systems that prevent condensation, and equipment designed without horizontal ledges or hidden cavities that collect condensation. These engineering decisions help determine whether water is likely to be uncontrolled in the environment in the first place. Engineering design choices help relieve the burden on behavioral interventions like wiping up water, reporting leaks, or adjusting cleaning practices due to facility design disadvantages. A plant that "designs water out" reduces its dependence on perfect human behavior and instead embeds moisture control into the physical structure of the facility itself.
Conclusion
Dry facilities have the advantage of a powerful safety barrier, but the introduction of water can erase that advantage by hydrating food residues, enabling microbial growth, damaging equipment, and undermining control systems designed for low-moisture environments. Academic research, industry experience, and recent FDA findings show that water introduction events can have significant consequences when they are not addressed. Ultimately, the most successful food safety programs are those that pair rigorous water control programs with engineering decisions that design water out of the environment altogether. Behavioral diligence will always matter, but a dry facility's strongest food safety protections come from a physical design that eliminates water.
Treating water like glass offers a practical framework for preventing these failures. The same principles that keep glass out of production by elimination where possible, strict control when necessary, and rapid response to deviations translate directly to water management. When facilities apply this mindset, water becomes an exception rather than an accepted background condition.
Acknowledgment
This work was supported in part by a grant from Dairy Management Inc. to Dr. Abby Snyder.
References
- U.S. Food and Drug Administration (FDA). " Call to Action Letter to the Powdered Infant Formula Industry." March 8, 2023. https://www.fda.gov/media/166044/download?attachment.
- FDA. "Warning Letter: JM Smucker LLC., MARCS-CMS 638042." January 24, 2023. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/jm-smucker-llc-638042-01242023.
- FDA. "Warning Letter: Guangzhou Sinocon Food Co., Ltd., MARCS-CSMS 692652." December 23, 2024. https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/guangzhou-sinocon-food-co-ltd-692652-12232024.
- Slaughter, C., S. Chuang, D. Daeschel, L. McLandsborough, and A.B. Snyder. "Moisture Matters: Unintended Consequences of Performing Wet Sanitation in Dry Environments." BioRxiv. https://www.biorxiv.org/content/10.1101/2025.11.26.690674v1.abstract.
- Grocery Manufacturers Association. "Water Audit Checklist for Low-Moisture Food Facilities." Snyder Lab at Cornell University. https://blogs.cornell.edu/snyder/tools-and-resources/.
Karl Thorson is the Food Safety and Sanitation Manager at General Mills. He was awarded the 2016 IAFP Sanitarian Award and provides leadership for the Cornell Dry Sanitation Advisory Council.
Abby Snyder, Ph.D. is an Associate Professor at Cornell University. She directs the Dry Sanitation Advisory Council, and her research group works with industry to understand and improve sanitation practices.

