FORMULATING WITH MIKE
Isocyanates, Part I
Fundamentals and Reactivity
Video credit: Hamster3d / Vetta via Getty Images
By Mike Praw, Senior Applications Scientist — Paints, Coatings and Inks, Indorama Ventures: Integrated Oxides and Derivatives, The Woodlands, TX
In the last column of Formulating with Mike, we covered self-leveling coatings. These coatings are often urethane-based. Since I have covered epoxies in previous columns, we will concentrate on urethanes. In the next few columns, we will cover isocyanates, polyols, and finally, some formulating tips for self-leveling coatings.
While monofunctional isocyanates exist, they are not generally used in the coatings space except as moisture scavengers. So, we generally either use diisocyanates or polymeric isocyanates. We normally refer to diisocyanates as monomeric isocyanates. Polyisocyanates, or polymeric isocyanates, have a functionality of over 2 — usually 3 or higher.
Isocyanates are generally separated into two broad types: aromatic and aliphatic, which describe their backbone. I will be concentrating on the main types of isocyanates. For aromatic isocyanates, methylene diphenyl diisocyanate (MDI) and toluene diisocyanate (TDI) are key. Due to the higher volatility and worse health profile of TDI versus MDI, TDI is not used in self-leveling coatings.
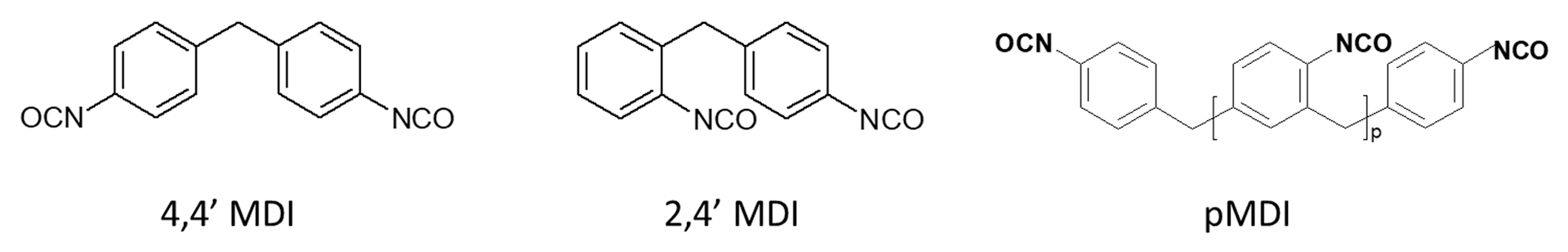
Table 1 shows isocyanate physical properties. From Table 1, TDI has a worse health rating and is 2,000 times more volatile, which precludes its use in most coatings. Structures of 2,4’ MDI, 4,4’ MDI, and PMDI are shown in Figure 1.
TABLE 1–ǀ–Isocyanate physical properties.
FIGURE 1–ǀ–MDI structures.
Isocyanates are powerful irritants to the membranes of the eyes, nose, throat, gastrointestinal, and respiratory tracts. One important factor about isocyanates is that some of them are allergic sensitizers, so proper personal protective equipment is critical, especially when spray-applying the coating. This generally precludes use by the general public of two-component urethane systems.
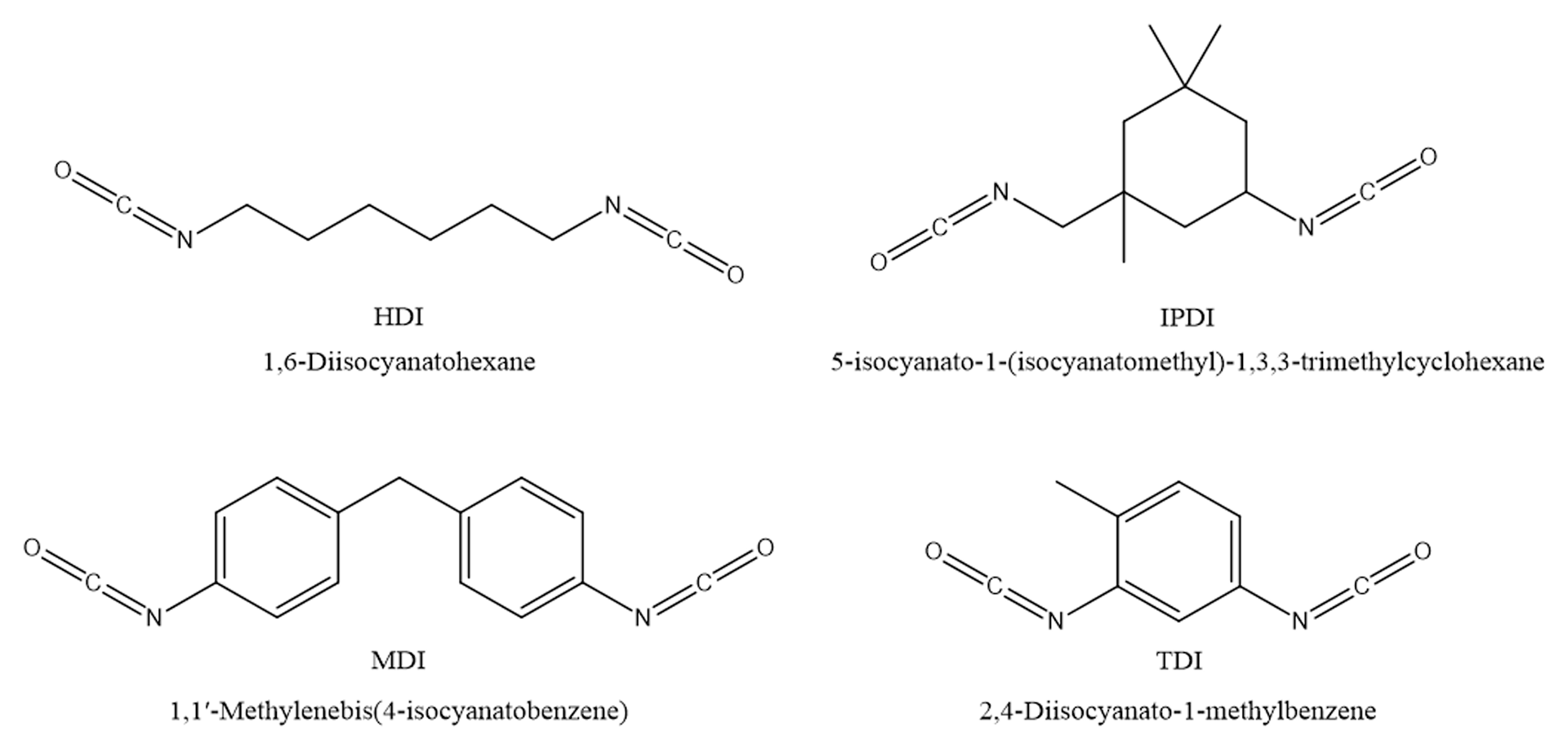
Aliphatic isocyanates have many advantages over aromatic isocyanates due to their low initial color and discoloration resistance in exterior applications. The most common aliphatic isocyanates are hexamethylene diisocyanate (HDI), methylene bis-cyclohexyl isocyanate — also known as hydrogenated MDI (H12MDI or HMDI) — and isophorone diisocyanate (IPDI). Hydrogenated MDI has the same structure as MDI, except that aliphatic cyclohexyl rings replace the aromatic rings.
MDI is relatively nonvolatile, while monomeric HDI is extremely volatile compared to MDI — 100,000 times more volatile. Monomeric IPDI is relatively nonvolatile but still too volatile to be used in this form. Isocyanate structures are shown in Figure 2.
FIGURE 2–ǀ–Diisocyanate structures.
Aromatic isocyanates are significantly faster reacting than aliphatic isocyanates. Normally, MDI will not need additional catalysts, while strong catalysts such as dibutyltin dilaurate are needed for aliphatic isocyanates. Relative reactivities of the first and second isocyanate groups are shown in Table 2.
TABLE 2–ǀ–Relative reactivity of the first and second isocyanate group with hydroxyl.
To combat the volatility of isocyanates, polymeric forms are produced. Not only are they safer to use, but the increased functionality of polymeric isocyanates raises the crosslink density of the coating (all other raw materials being equal). This leads to better chemical resistance and higher hardness but can decrease flexibility.
Polymeric aliphatic isocyanates (small “p” before the isocyanate acronym) are constantly evolving to meet the new demands of the coatings industry. While exact numbers are hard to find, general consensus is that polymeric HDI accounts for 50%-60% of all aliphatic isocyanate sales in North America.
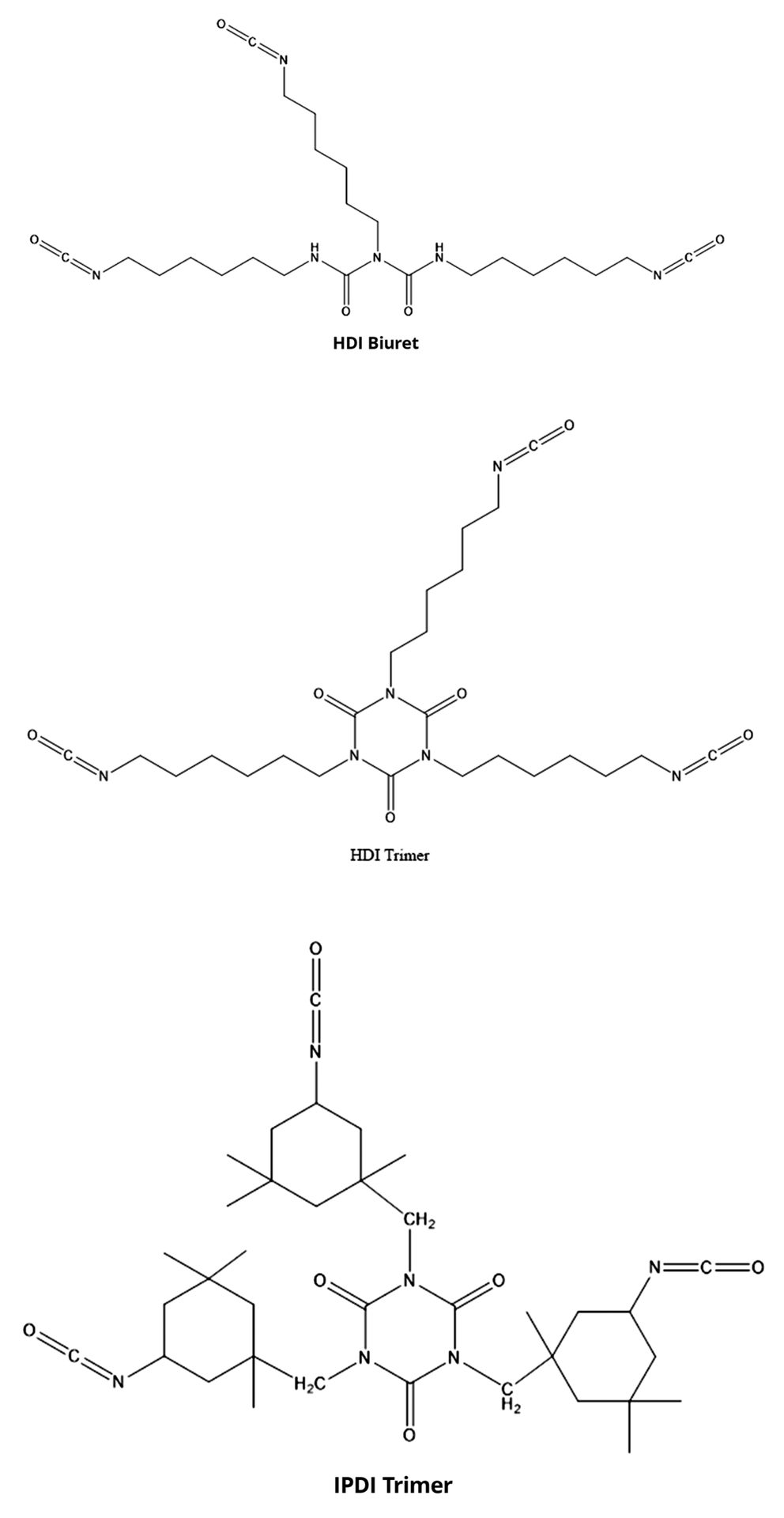
For pHDI, the initial form was supplied as a biuret. While its properties were good, its high viscosity (5,000-10,000 centipoise) required it to be supplied cut in solvent. As lower volatile organic content (VOC) versions were needed, HDI trimer versions were produced. Finally, as lower viscosity became the norm, allophanate structures were developed. Sample structures of polymeric aliphatic isocyanates are shown in Figure 3.
FIGURE 3–ǀ–Aliphatic isocyanate structures.
One major advantage of isocyanates is that they react with both hydroxyl (a in Figure 4) and amine groups (c in Figure 4), with the amine reaction being significantly faster — by orders of magnitude. However, since they react with hydroxyl groups, they also react with water. This reaction (b in Figure 4) leads to CO₂ formation, which causes bubbles in coatings, leading to defects. A little water goes a long way. One molecule of water will react with an isocyanate to form an amine (b in Figure 4), and then a second isocyanate group will react with the amine (c in Figure 4). So, 18 g of water will react with 168-250 g of monomeric isocyanate, generating significant amounts of CO₂.
FIGURE 4–ǀ–Isocyanate reactions.

Because of this, it is critical that the coating contains raw materials with little or no water. Many pigments contain significant water — for example, up to 8% by weight for calcium carbonate. Once the coating is made, moisture scavengers are recommended for the paint side of the two-part system. Zeolite powders are common moisture scavengers for pigmented systems. For clear systems, monomeric isocyanates or silanes, such as vinyltrimethoxy silane, are commonly used.
Another issue from this water reaction is that the substrate must be dry. This means that applying urethanes over raw concrete is not possible, and applicators must be aware of the dew point and temperature of the substrate. While the temperature may be warm, if the concrete is cooler, condensation will occur. This becomes even more complicated in solvent-based systems because solvent evaporation can cool the coating below the dew point, leading to water condensation on the surface.
Because some water will always be present, it is recommended to add 5%-10% excess isocyanate to coatings formulations to compensate for urea formation. Excess unreacted polyol will harm film properties more than some additional urea. Excess isocyanate will moisture cure over time and have minimal effect on film properties.
Using Isocyanates in Water-Based Systems
How do we use isocyanates in water-based systems? There are a couple of methods to protect the NCO group from water.
The first is to emulsify the isocyanate using a surfactant. This colloidal protection prevents the water outside the micelle from reacting with the isocyanate group inside the micelle.
The second method is to block the isocyanate so that its reaction is slowed. This way, by the time it has started reacting, most of the water has evaporated. Neither of these methods fully prevents water-isocyanate reactions, so it is normal to add excess isocyanate — often two to three times the stoichiometric amount — to ensure adequate crosslinking. The excess isocyanate reacts with water, forming urea.
In the next column, we will cover the components that react with isocyanates.
All information contained herein is provided "as is" without any warranties, express or implied, and under no circumstances shall the author or Indorama be liable for any damages of any nature whatsoever resulting from the use or reliance upon such information. Nothing contained in this publication should be construed as a license under any intellectual property right of any entity, or as a suggestion, recommendation, or authorization to take any action that would infringe any patent. The term "Indorama" is used herein for convenience only, and refers to Indorama Ventures Oxides LLC, its direct and indirect affiliates, and their employees, officers, and directors.
